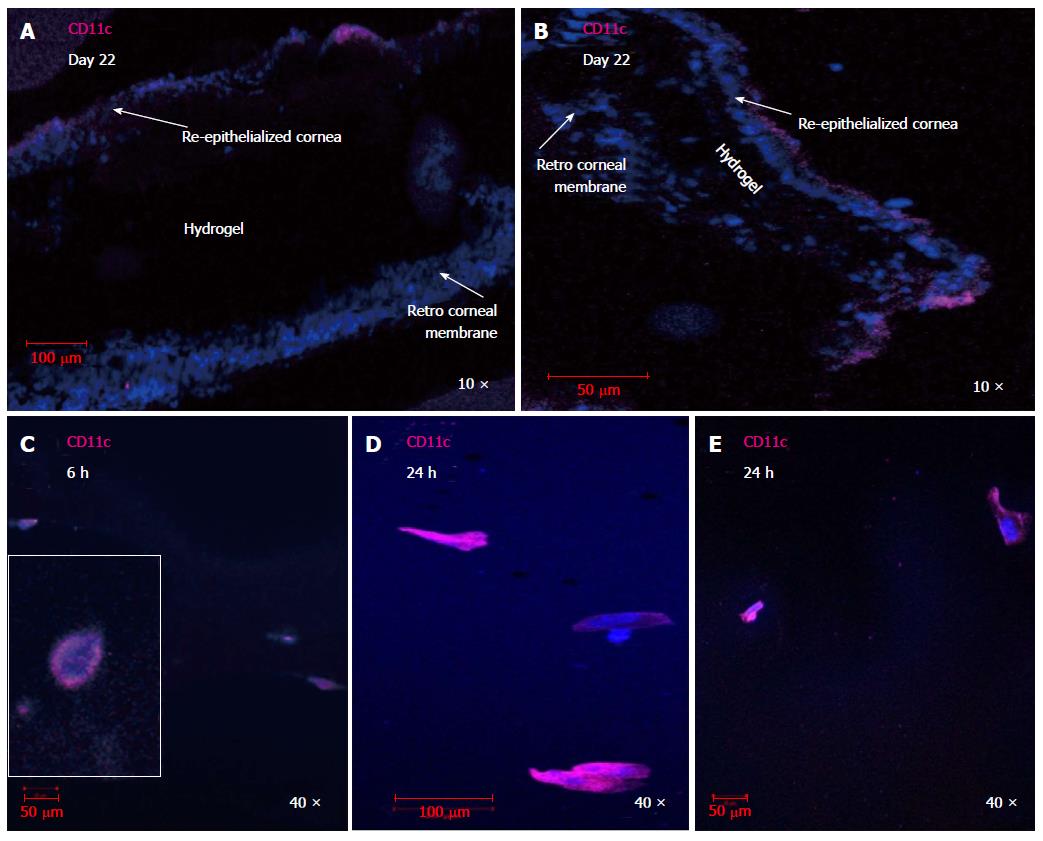
Unbiased profiling of Sp7 target genes and binding sites in osteocytes reveals a stage-specific role for this transcription factor during osteocytogenesis. 
In this work, we delete Sp7 in mature osteoblasts and osteocytes and observe a dramatic skeletal phenotype including cortical porosity, increased osteocyte apoptosis, and severe defects in osteocyte dendrites. However, the function of Sp7 in osteocyte maturation remains unknown. Common human SP7-associated variants are linked to bone mineral density variation and fracture risk 12, 13, and rare SP7 mutations cause recessive forms of osteogenesis imperfecta 14, 15. Osterix/Sp7 (encoded by the Sp7 gene) is a zinc finger-containing transcription factor essential for osteoblast differentiation and bone formation downstream of Runx2 11. The goal of this study was to define key gene regulatory circuits that drive osteocyte differentiation and dendrite formation. Lineage-specifying transcription factors have been identified for other key cell types in bone 10 however, lineage-defining transcription factors that coordinate genetic programs associated with osteocyte maturation remain unknown. Recent estimates suggest that the osteocyte connectivity network in human bone exhibits the same order of complexity as the network of connections between neurons in the brain 9. Defects in the osteocyte dendrite network may cause skeletal fragility in the setting of aging and glucocorticoid treatment 7, 8. The osteocyte dendritic network confers mechano-sensitivity to these cells and allows for extensive communication among osteocytes and adjacent cells on bone surfaces 6. Furthermore, osteocytes have a unique morphology as they bear multiple long, neuron-like dendritic processes projecting through the lacunar canalicular system in bone 5. These cells translate external cues, such as hormonal variations and mechanical stresses, into changes in bone remodeling by secreting paracrine-acting factors that regulate osteoblast and osteoclast activity 4. Recently, emerging evidence has highlighted key roles for osteocytes in bone remodeling 3. Although the roles of osteoblasts and osteoclasts in bone formation and resorption have been well studied 1, 2, those of osteocytes, the most abundant cell type in bone, had been overlooked due to technological limitations and the cells’ relatively inaccessible location within the mineralized bone matrix. The major cell types that govern bone homeostasis are osteoblasts, osteoclasts, and osteocytes. These findings reveal a role for Sp7 and its target gene Osteocrin in osteocytogenesis, revealing that pathways that control osteocyte development influence human bone diseases. Sp7-dependent genes that mark osteocytes are enriched in neurons, highlighting shared features between osteocytic and neuronal connectivity. Moreover, humans with a SP7 R316C mutation show defective osteocyte morphology. Sp7-dependent osteocyte gene networks are associated with human skeletal diseases. Single-cell RNA-sequencing demonstrates defects in osteocyte maturation in the absence of Sp7. Osteocrin is a Sp7 target gene that promotes osteocyte dendrite formation and rescues defects in Sp7-deficient mice. Profiling of Sp7 target genes and binding sites reveals unexpected repurposing of this transcription factor to drive dendrite formation. Here we show that deletion of Sp7 in osteoblasts and osteocytes causes defects in osteocyte dendrites. The circuitry that drives dendrite formation during “osteocytogenesis” is poorly understood. Some osteoblasts embed within bone matrix, change shape, and become dendrite-bearing osteocytes. Nature Communications volume 12, Article number: 6271 ( 2021) Control of osteocyte dendrite formation by Sp7 and its target gene osteocrin
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
